Can You Trust your Biological Samples?

Our friends at Lab Manager recently published an article written by Arizona State University that stressed the importance of sample integrity when conducting scientific research. Millions of dollars are spent each year to conduct research that seeks to cure debilitating diseases. This often involves studying biological samples to discover appropriate biomarkers and subsequently developing treatments for disease. Thus the foundation of successfully treating diseases lies in the integrity of these biological samples. But biology is fussy - all living things require certain environmental conditions (such as the right temperature, humidity, atmospheric gas composition, etc.) to live and thrive. That’s why it’s important to monitor these conditions when carrying out scientific research. We’ve come across so many stories of improper sample storage conditions. These typically don’t happen due to negligence, but more often due to circumstances beyond our control. One example is the California power outages, and another is faulty cold storage equipment.
Shifts in environmental conditions as well as fluctuations in the collection, processing, storage, and handling of samples can compromise experimental outcomes. After all, we’ve all heard the term "Garbage in, Garbage out". In fact, this is one of the contributors behind experimental irreproducibility, a phenomenon that has been gaining more and more attention in recent years. Experimental irreproducibility costs billions of dollars in lost productivity and hinders scientific advancement. Thirty-six percent (36%) of irreproducible results can be attributed to biological reagents and materials issues. See Figure 1. The Lab Manager article highlights an interesting point from Chad Borges of the Biodesign Virginia G. Piper Center for Personalized Diagnostics at Arizona State University: “One of the major reasons that there are so many discoveries of biomarkers (early indicators of disease) in the literature, but so few positive validations that confirm those findings is the fact that in many cases during the discovery, samples were used that have a history or an integrity that's simply unknown." He has even experienced the pain himself when he observed major differences between the two sets of samples for a specific biomarker. He ultimately learned that the freezer in which the control samples were stored had lost power for several days during a natural disaster. That piece of information was the key to explaining the differences between the two sets of samples.
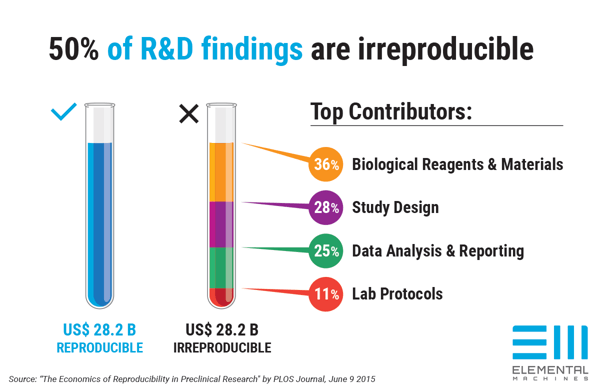
Figure 1: Top contributors of irreproducible R&D results sum up to $28 billion in losses
One of the easiest ways to safeguard sample integrity is through 24/7 comprehensive environmental monitoring. Are we suggesting installing a
complex, hardwired monitoring solution? Absolutely not! Systems like the Elemental Machines Connected Monitoring Platform run off of new technology such as the Internet of Things (IoT) and Cloud storage, are easily deployable, and gather continuous information about your samples. User friendly interfaces make it easy to understand whether your samples are safe or whether there could be a problem. It’s also a good idea to have a system that is equipped with battery and cellular backup, so that it keeps working in the event of power and/or wifi outages. This provides lab directors and researchers with peace of mind knowing their samples are protected.

Figure 2: The Elemental Machines Connected Monitoring Platform uses Element-A and Element-T to collect data from lab equipment and environments

Of course, monitoring is only half the solution. The system must be able to alert the responsible team members before any samples are compromised. Some systems use audible alarms, which are fine if someone is present in the laboratory to hear them. Audible alarms are not particularly useful at night or on the weekends if nobody is working. A much better solution is to send email and text alerts to a designated team of people in the event of out-of-spec conditions. Another nice feature is having the ability to document actions taken to resolve the alert situation. These notes can be particularly useful for 21 CFR Part 11 compliance or other regulatory codes. This type of system provides the most comprehensive coverage for verifying sample integrity, troubleshooting experimental reproducibility, and supporting regulatory audits. To learn more about how the Elemental Machines Platform can help organizations like yours, contact us to request a demo of our solution.
Never miss a beat...
Subscribe to our bi-monthly EM Connect Newsletter to stay up-to-date on all things LabOps, biotech, and more!
